
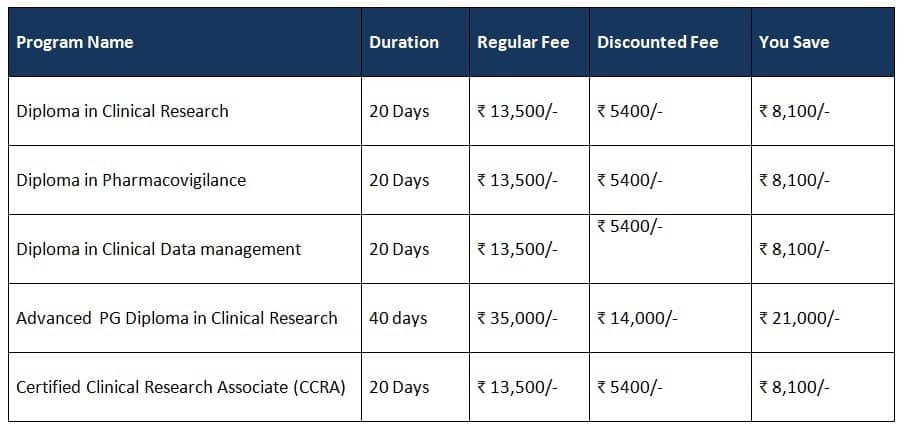
Program Details :
This is a foundation educational program, designed for all the candidates aspiring to work in the field of clinical research or allied professions like central labs. The program provides complete overview and practical environment in the field of clinical research. The program would allow candidates to upgrade their knowledge about ICH GCP Guidelines, regulatory issues and other major aspects of clinical trials management. Select important topics from the following groups would be covered in the program:
* Clinical Research Introduction
* Roles & Responsibilities of Key Stakeholders
* Preparations & Planning for Clinical Trials
* Essential Documentation in Clinical Research & Regulatory Submissions
* Clinical Trials Project Planning & Management
* Study Start Up Process
* Clinical Monitoring Essentials
* Compliance, Auditing & Quality Control in Clinical Research
Course Objective :
* To provide a comprehensive introduction to the clinical research process.
* Learn the skills, knowledge and competencies of a candidate for the Clinical Research Associate / Clinical Research
* Coordinator position.
* Become more familiar with roles/jobs as part of the study team.
Inclusions:
• Training Session by Certified Industry Subject Matter Experts.
• Globally recognized Program Content and Course Materials.
• Case Studies.
• No Examination processing charges.
• Practical exposure on CDM and Pharmacovigilance tools.
• Globally recognized Program Certification.
• 100% Placement Assistance.
• Resume Development.
• Soft Skills / Communication Training.
• Interview Preparation / Pre-recruitment training.
• All Necessary Stationary.
• Exclusive Discount Vouchers for future Learning.
• Referral support for quick placements.
• PharmaExa Goodies and more